
4)Atoms of different elements combine in simple whole number ratios. He expected the particles to go straight through like in the plum pudding model, but even though most went through, some bounced back. 3) Chemical Compounds are formed by the union of two or more atoms of different elements. Atomic theory quiz 4.0 (1 review) Get a hint The modern model of the atom describes electrons in a little less specific detail than earlier models did. Study with Quizlet and memorize flashcards containing terms like Democritus, Law of Conservation of Matter, Law of definite proportion and more. 2)Atoms of the same element are same in mass and size. Therefore, while the Law of Definite Proportions can be used to compare two experiments in which hydrogen and oxygen react to form water, the Law of Definite Proportions can not be used to compare one experiment in which hydrogen and oxygen react to form water, and another experiment in which hydrogen and oxygen react to form hydrogen peroxide (peroxide is another material that can be made from hydrogen and oxygen). Terms in this set (26) Daltons Atomic Theory (5 points) 1) Matter is made up of tiny, indivisible, indestructible atoms. The Law of Definite Proportions applies when elements are reacted together to form the same product. Law of Definite Proportions states that in a given type of chemical substance, the elements are always combined in the same proportions by mass. the particles of the nucleus that have no charge. center of an atom positively charged made up of protons and neutrons. 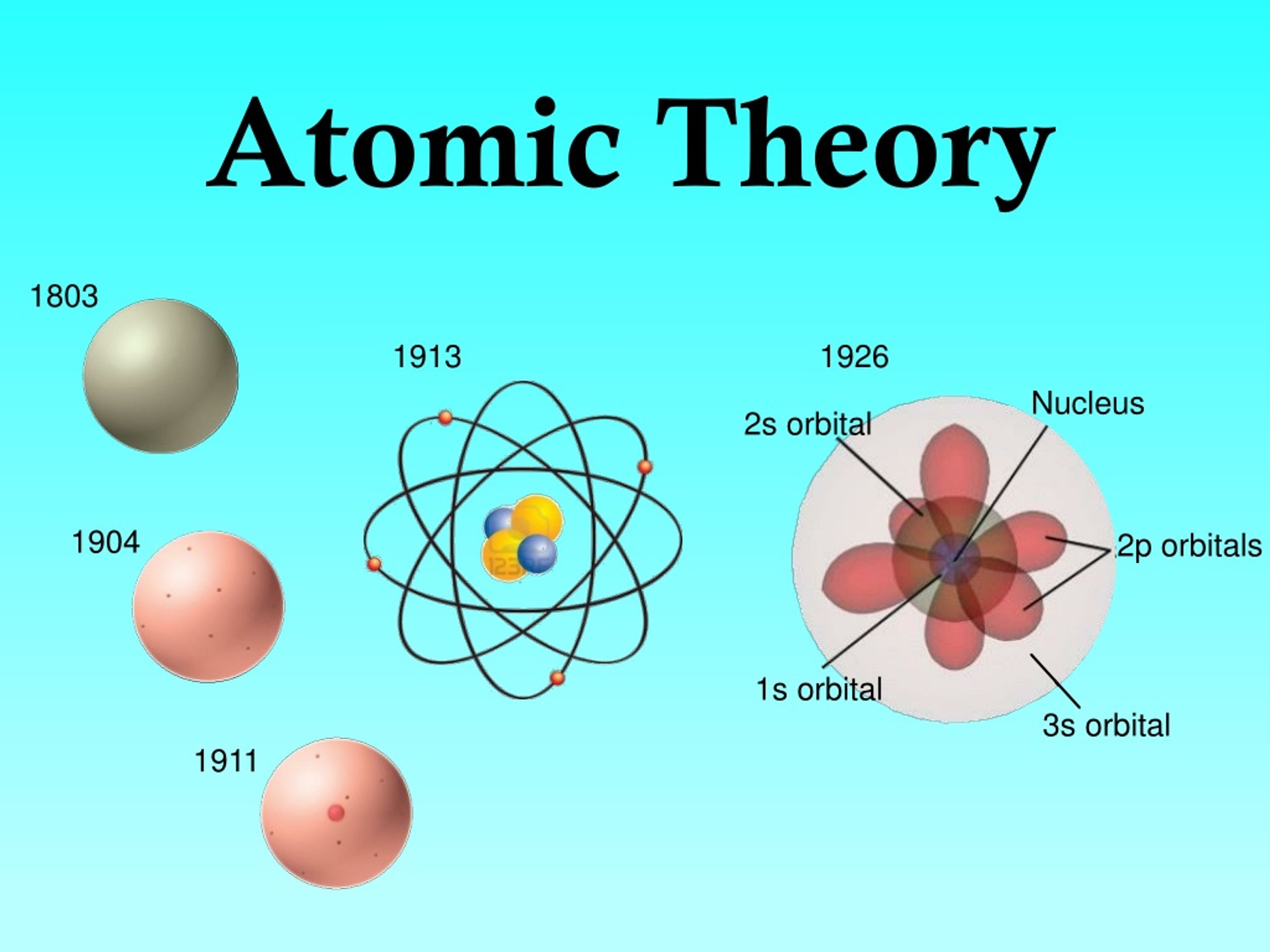
Similarly, when 2 grams of A react with 16 grams of B, they must produce 18 grams of C. a theory that states that all matter is composed of tiny particles called atoms. All elements are composed of tiny individual particles called atoms (subatomic particles proved this to. Which parts of Daltons atomic theory had to be revised 1. If 1 gram of A reacts with 8 grams of B, then by the Law of Conservation of Mass, they must produce 9 grams of C. Study with Quizlet and memorize flashcards containing terms like Who used the plum pudding model, Who used the gold foil experiment, Who used the oil drop experiment and more. Atoms combine in single, whole number ratios to form compounds. Atoms are neither created nor destroyed in chemical reactions. \) summarizes the properties of these three subatomic particles.\): If 1 gram of A reacts with 8 grams of B, then by the Law of Definite Proportions, 2 grams of A must react with 16 grams of B. Study with Quizlet and memorize flashcards containing terms like Who and when first proposed that matter consists of atoms, What were the properties of atoms according to those who introduced them, How a solid transforms into a liquid according to the 'atomists,' those who believed that matter is made of atoms and more. Study with Quizlet and memorize flashcards containing terms like The first model of the atom was developed through, The model of the atom has changed as scientists have gathered new evidence. Atoms of a given element are identical to one another.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
